Study of some reactions of proteins
Proteins are complex macromolecules composed of amino acids linked by peptide bonds. Their specific three-dimensional structure is maintained by hydrogen bonds, ionic interactions, hydrophobic interactions, and disulfide bridges. This ordered structure (primary, secondary, and tertiary levels) is essential for protein functionality.
When proteins are exposed to heat, these stabilizing interactions weaken and eventually break. This process is known as denaturation. During denaturation, the protein unfolds from its native conformation, as shown in figure below. Although the primary structure (peptide bonds) remains intact, the secondary and tertiary structures are disrupted. As heating continues, unfolded protein molecules interact with each other, forming aggregates. This aggregation leads to coagulation and visible solidification.
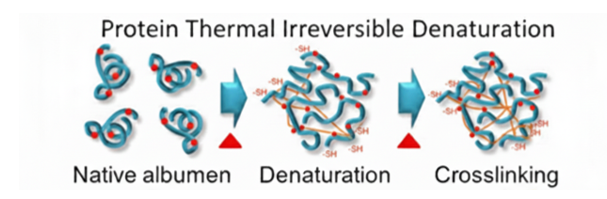
Egg albumen, mainly composed of ovalbumin, is an ideal model system to demonstrate protein denaturation. At room temperature, egg white appears translucent and fluid because proteins are in their native folded state. Upon heating, denaturation begins (around 60–65°C), and progressive coagulation occurs. At higher temperatures, a firm, opaque gel is formed due to extensive protein aggregation.
The following figure represents how the egg albumen turns from liquid to solid upon heating due to protein denaturation

This temperature-dependent transformation clearly demonstrates how heat affects protein stability and structure, making the process of denaturation visually observable in egg albumen.
