Quantum Nature of Light with Young’s Double-Slit Experiment
Wave Nature of Light
Light exhibits a dual nature — it behaves both as:
- Particle (photon)
- Wave (electromagnetic wave)
When light interacts with obstacles or openings comparable to its wavelength (λ), it bends around the edges. This phenomenon is known as diffraction.
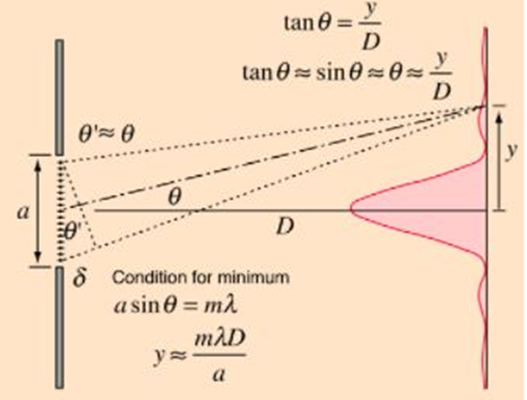
Fig. 1 Diffraction Pattern by Single-Slit
Phenomena explained through the wave nature of light:
- Reflection
- Refraction
- Dispersion
- Diffraction
- Interference
- Polarization
1. Single-Slit Diffraction
When monochromatic light passes through a narrow slit of width a, it spreads out and forms a diffraction pattern on the screen.
(i) Condition for Minima (Dark Fringes)
a · sinθ = mλ (m = ±1, ±2, ±3, …) — (1)
Where:
- a → slit width
- λ → wavelength of light
- θ → angular position of minima
- m → diffraction order
(ii) Linear Fringe Position on Screen
If the screen is at distance D from the slit:
y = mλD / a — (2)
(iii) Key Observations
- Central maximum: Brightest and twice as wide as other maxima.
- Side maxima: Much weaker and decrease in intensity.
- Higher-order maxima: Spread further apart as m increases.
- Effect of Slit/Wavelength:
- Narrower slit → wider diffraction pattern.
- Longer wavelength → greater spreading.
Note: This experiment demonstrates the wave nature of light.
2. Double-Slit Interference (Young's Experiment)
When coherent light passes through two slits separated by distance d, the waves overlap and interfere.
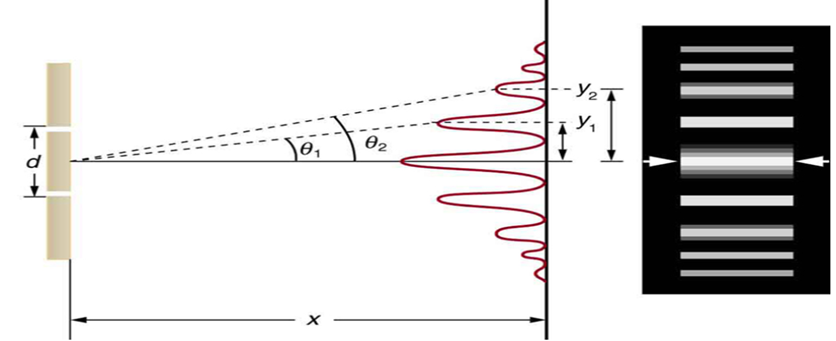
Fig. 2 Young's double slit interference
1. Fringe Width
β = λx / d — (3)
Where:
- β → fringe width
- λ → wavelength
- x → screen distance
- d → distance between slits
2. Interference Pattern
Bright Fringes (Constructive Interference)
Δ (Path difference) = nλ — (4)
Dark Fringes (Destructive Interference)
Δ (Path difference) = (2n + 1) · λ/2 — (5)
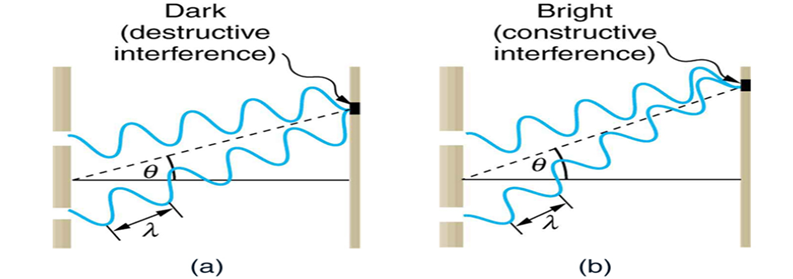
Fig. 3 Destructive and constructive interference
3. Significance
- Confirms the wave nature of light.
- Supports Huygens' Principle.
- Demonstrates superposition of waves.
3. Electron Double-Slit Experiment (Quantum Theory)
This modern experiment shows that matter also behaves like waves (matter waves or de Broglie waves).
1. Key Observations
- Electrons fired one at a time still produce an interference pattern, which shows electrons behave like probability waves.
- If we measure which slit the electron passes through (with detector):
- The interference pattern disappears.
- Only two humps remain (each corresponding to one slit-classically expected).
- The electron behaves like a classical particle.
- Measurement collapses the wave function.
Wave–Particle Duality:
- Electrons behave like particles when detected.
- But behave like waves when propagating.
2. Implications
- Supports de Broglie hypothesis.
- Demonstrates quantum superposition.
- Shows observer effect / wave function collapse.
- Reveals that nature at the microscopic level is probabilistic, not deterministic.
