Determination of rate/ extent of hydrolysis of sucrose/starch
The hydrolysis of sucrose and starch involves the breakdown of complex carbohydrates into simpler sugars through chemical or enzymatic reactions. Understanding these reactions is important in food processing, brewing, confectionery, and bioethanol production.
- Hydrolysis of Sucrose (Acid-Catalyzed Inversion)
Sucrose is a disaccharide composed of glucose and fructose. In the presence of dilute acid (e.g., HCl) and heat, sucrose undergoes hydrolysis (inversion) to produce an equimolar mixture of glucose and fructose, known as invert sugar.
Reaction Equation:
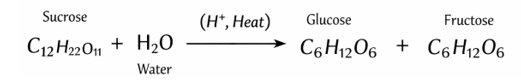
This reaction increases reducing sugar content because both glucose and fructose are reducing sugars. Monitoring this reaction is important where sweetness, fermentability, and stability of syrups are critical. - Hydrolysis of Starch (Enzymatic Action)
Starch is a polysaccharide composed of amylose and amylopectin linked by α-1,4 and α-1,6 glycosidic bonds. Enzymatic hydrolysis is carried out using α-amylase, which cleaves α-1,4 bonds, producing maltose, dextrins, and glucose.
Simplified Reaction:
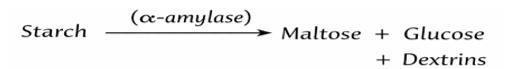
This process converts non-reducing polysaccharides into reducing sugars. - Estimation of Reducing Sugars (Fehling’s Test)
The extent of hydrolysis is determined by estimating reducing sugars using Fehling’s solution.
Reaction Principle:

Blue Cu²⁺ ions are reduced to brick-red copper(I) oxide (Cu₂O), indicating the presence of reducing sugars. The volume of sample required to reduce a known volume of Fehling’s solution allows calculation of reducing sugar concentration, which correlates with the degree of hydrolysis.
This experiment helps in understanding chemical versus enzymatic hydrolysis, the role of acids and enzymes in glycosidic bond cleavage, and the quantitative estimation of reducing sugars to evaluate reaction rate and extent of hydrolysis.
