1. Determination of heat stability of vitamin C
Vitamin C (ascorbic acid) is an essential water-soluble vitamin widely present in citrus fruits and fruit juices. However, it is highly sensitive to heat, oxygen, light, and metal ions. During thermal processing operations such as pasteurization and sterilization, significant degradation of Vitamin C may occur. This degradation affects both nutritional quality and labeling accuracy of processed foods.
Ascorbic acid undergoes oxidation to form dehydroascorbic acid, which can further degrade into biologically inactive compounds. The degradation rate increases with temperature; therefore, monitoring Vitamin C concentration at different heating temperatures (60°C, 75°C, and 90°C) helps evaluate thermal stability and nutrient retention during processing.
Reaction Principle
The determination of Vitamin C in this experiment is carried out using the 2,6-dichloroindophenol (DCPIP) titrimetric method.
Ascorbic acid (reducing agent) reduces the blue dye (DCPIP) to a colorless form:
Ascorbic Acid (reduced form) + DCPIP (blue) → Dehydroascorbic Acid + Reduced DCPIP (colorless)
At the endpoint, when all Vitamin C is oxidized, the first excess drop of unreduced dye imparts a light rose-pink color, indicating completion of titration.
The dye is first standardized using a known concentration of standard ascorbic acid solution.
The titre value obtained is then used to calculate Vitamin C content in heated juice samples.
Process Overview (Flow Chart)
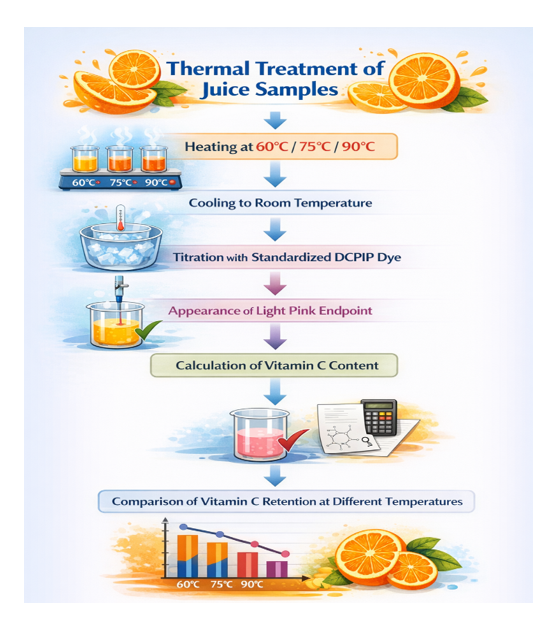
By comparing the titration values of samples treated at different temperatures, the stability
trend of Vitamin C during thermal processing can be evaluated. Higher temperatures are generally associated with greater Vitamin C loss due to accelerated oxidation.
