Chemical composition of cement
Preparation of Cement
Step 1: Click on the trowel to transfer cement onto the crucible.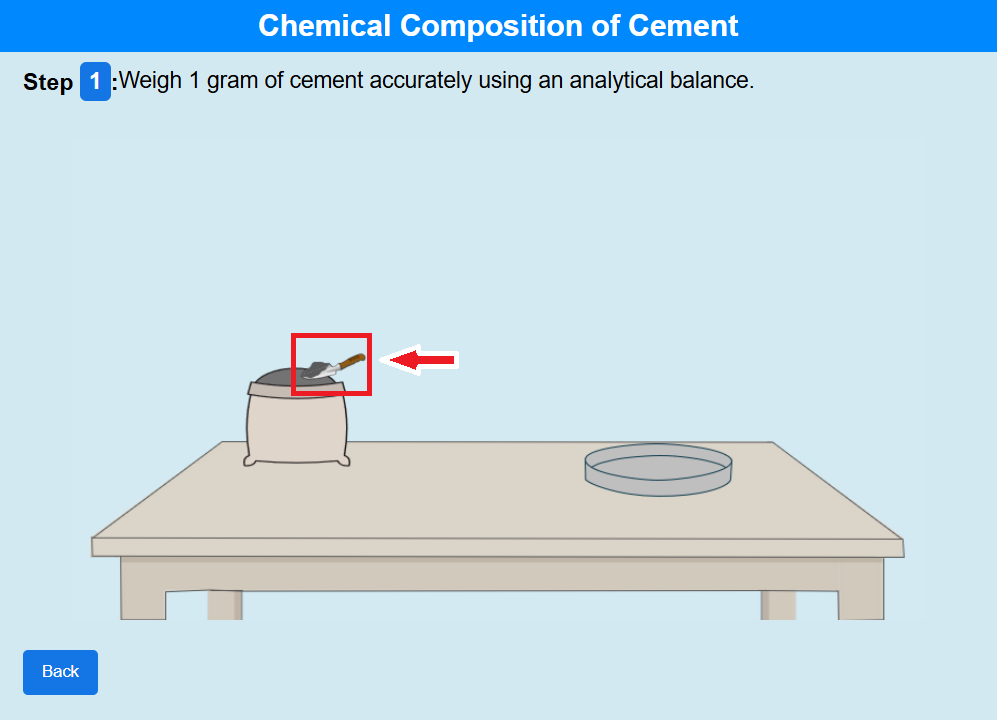
Step 2: Click on the towel to weigh 1 gram of cement accurately using an analytical balance.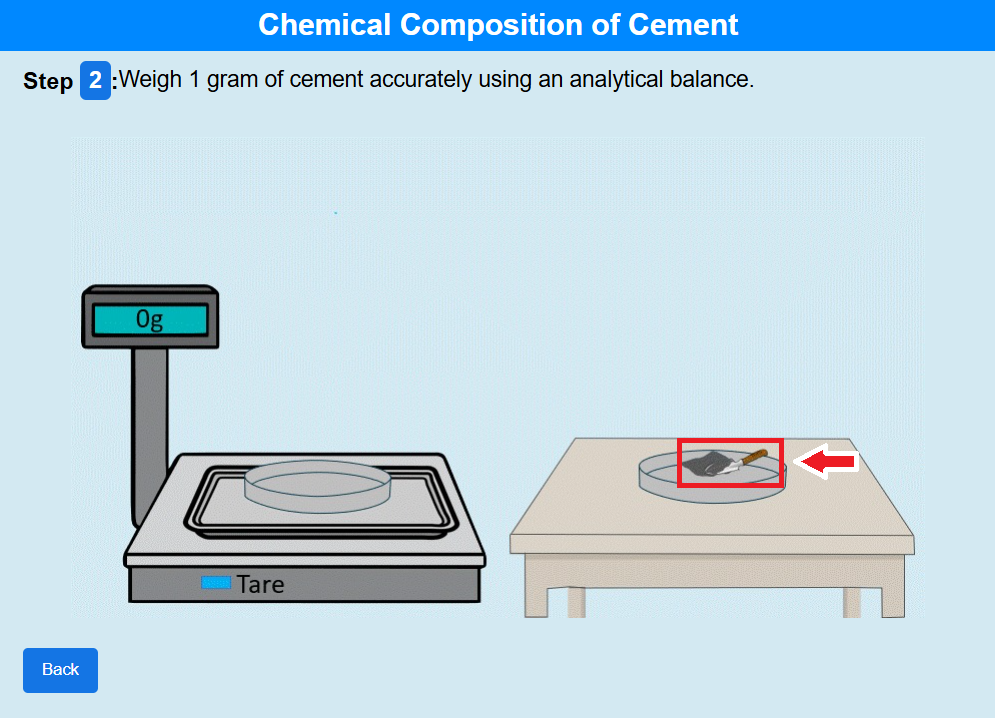
Step 3: Click on the crucible to transfer the cement sample to a beaker.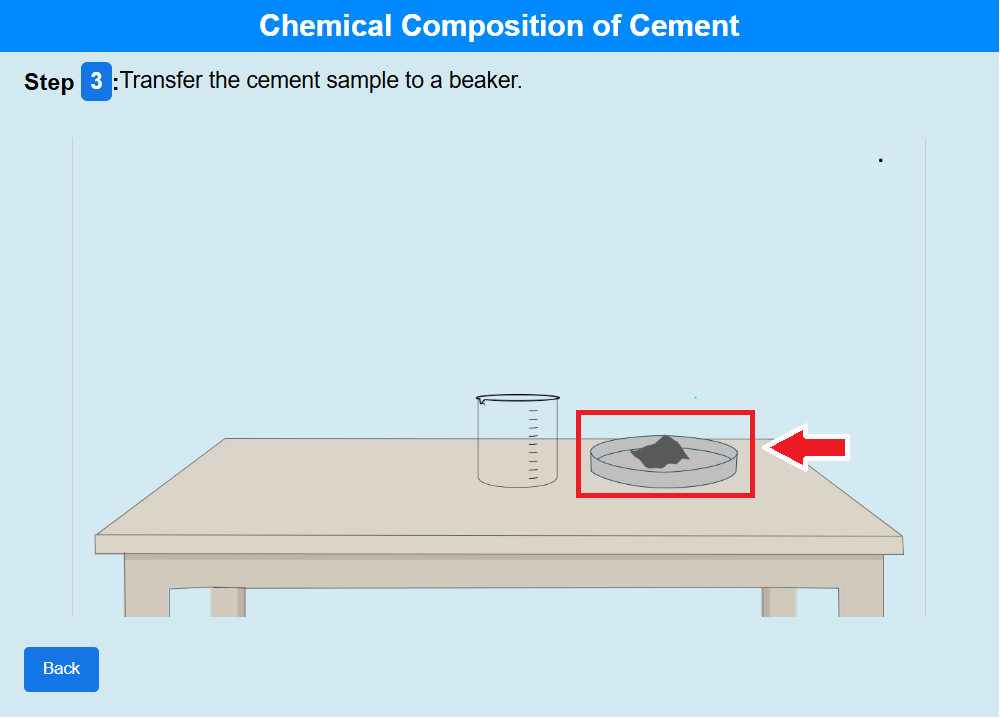
Step 4: Click on the HCl bottle to add 20 mL of dilute hydrochloric acid (HCl) to the beaker.
Step 5: Click on the beaker to heat the solution gently.
Step 6: Click on the Funnel to filter the solution to remove any undissolved particles.
Step 7: Click on the measuring cylinder to transfer the filtered solution to a clean container for further analysis.
Determination of Calcium Oxide
• Click on the link to perform the test of Determination of Calcium Oxide.
Step 1:
• Click on the pipette to add ammonium hydroxide (NH₄OH) dropwise to the filtrate until
The solution is slightly alkaline (pH approximately 8–9).
Step 2:
• Click on the measuring cylinder to add 10 mL of ammonium oxalate ((NH₄)₂C₂O₄) solution to
precipitate calcium oxalate.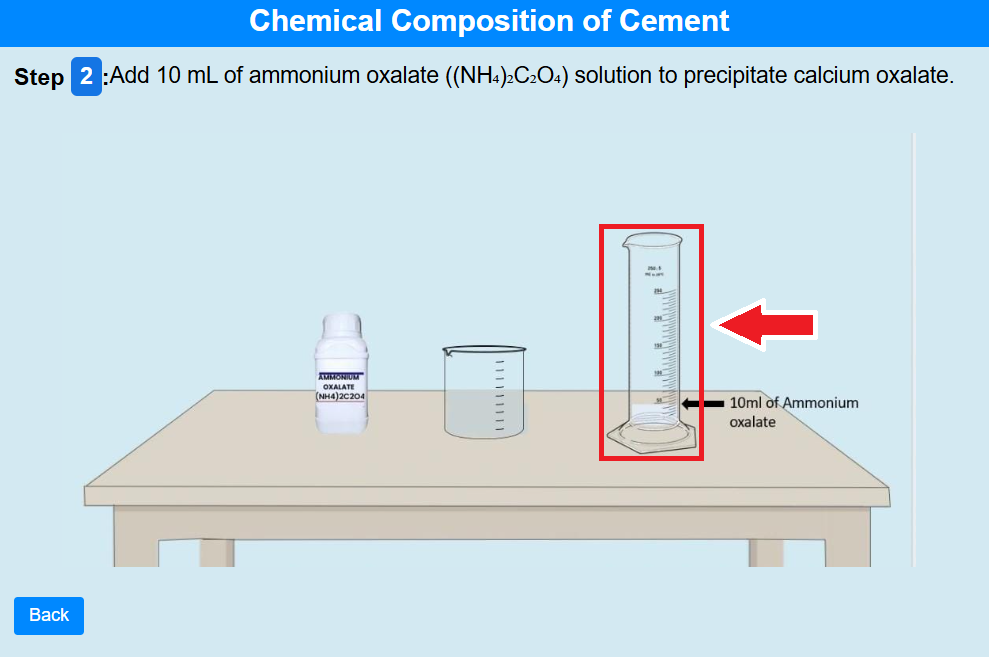
Step 3:
• Click on the filter paper to filter the precipitate.
• Click on the knob of the oven to open it.
• Click on the crucible to dry it in an oven at 110°C.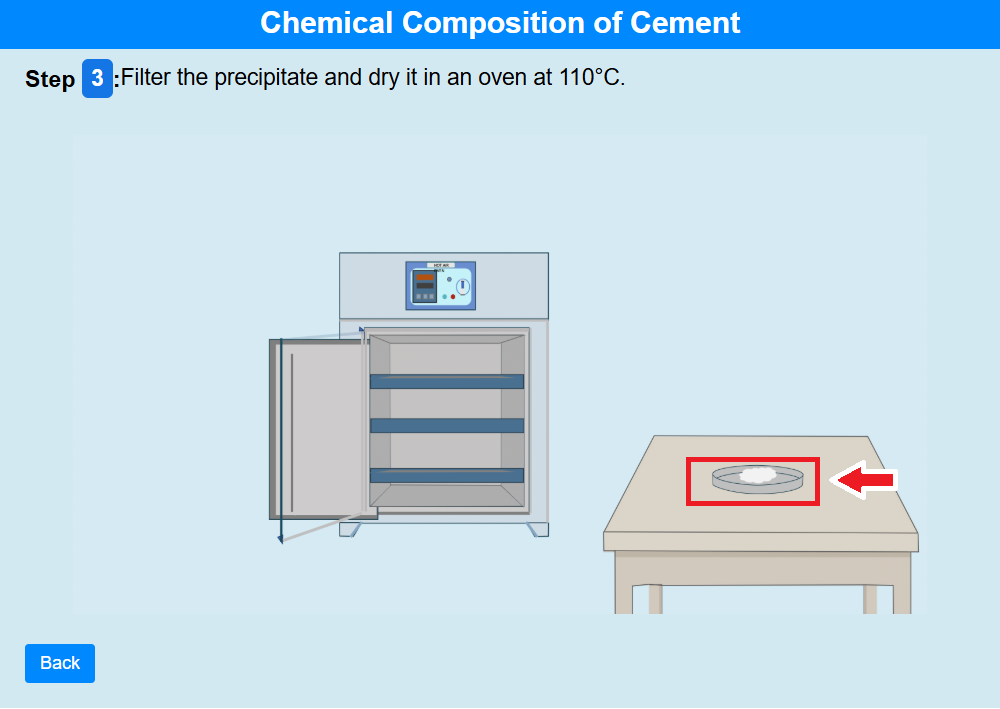
• Click on the screen to set the temperature to 110°C to dry.
Step 4:
• Click on the crucible to weigh the dried precipitate and calculate the percentage of CaO.
• Click on the calculate CaO % to calculate the percentage of CaO.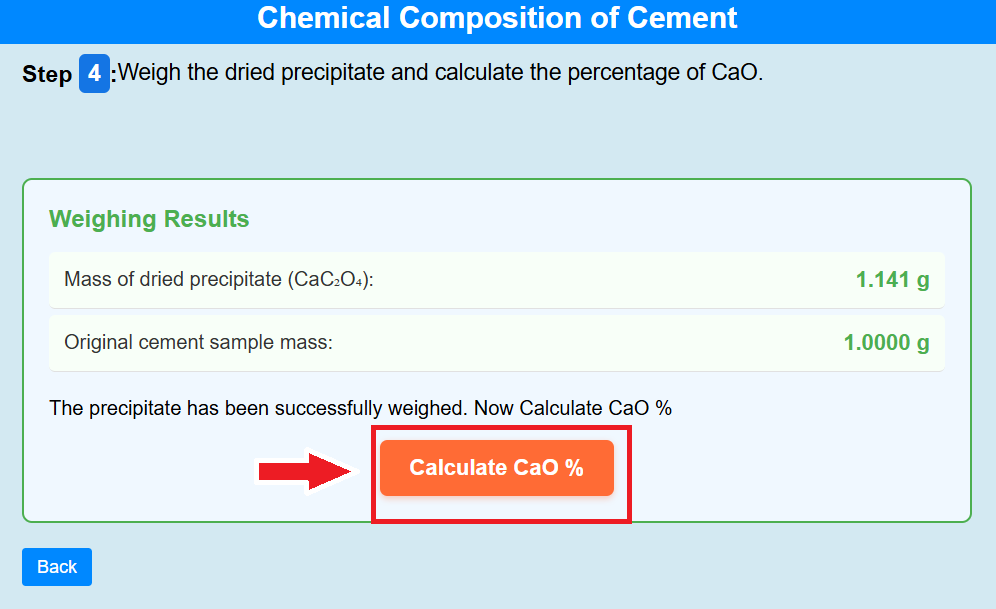
Determination of Iron Oxide and Aluminum Oxide
• Click on the link to perform the test of Determination of Iron Oxide and Aluminium Oxide.
Step 1:
• Click on the pipette to another portion of the filtrate, and add ammonium hydroxide (NH₄OH)until a precipitate forms.
Step 2:
• Click on the filter paper to filter the precipitate, which contains ferric hydroxide [Fe(OH)₃] and aluminum hydroxide [Al(OH)₃].
Step 3:
• Click on the measuring cylinder to dissolve the precipitate in dilute hydrochloric acid (HCl).
• Click on the crucible to dissolve the precipitate in dilute hydrochloric acid (HCl).
Step 4:
• Click on the pipette to add a few drops of nitric acid (HNO₃) to the solution.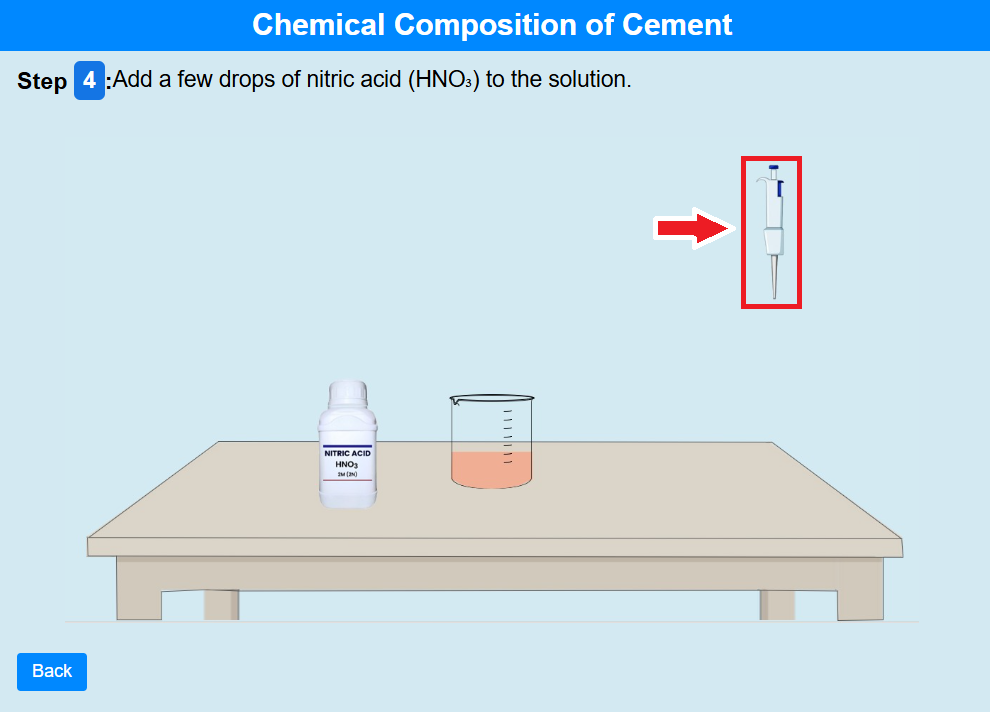
Step 5:
• Click on the pipette to add dilute sulfuric acid (H₂SO₄) to provide the required acidic medium.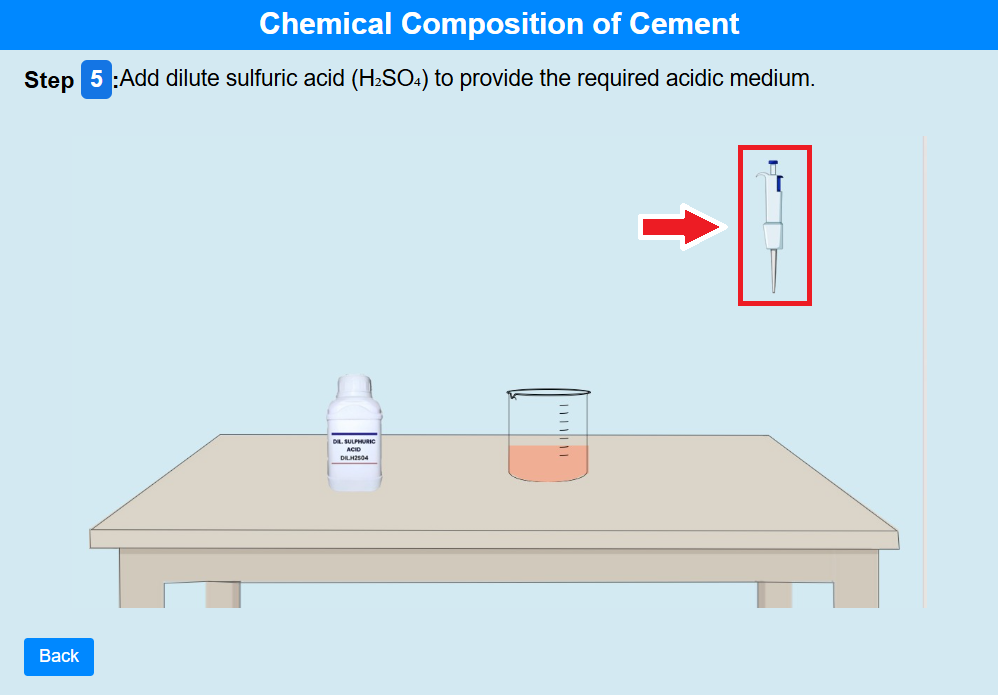
• Click on the beaker to transfer the solution to a conical flask and titrate with potassium
permanganate (KMnO₄) to determine iron oxide (Fe₂O₃).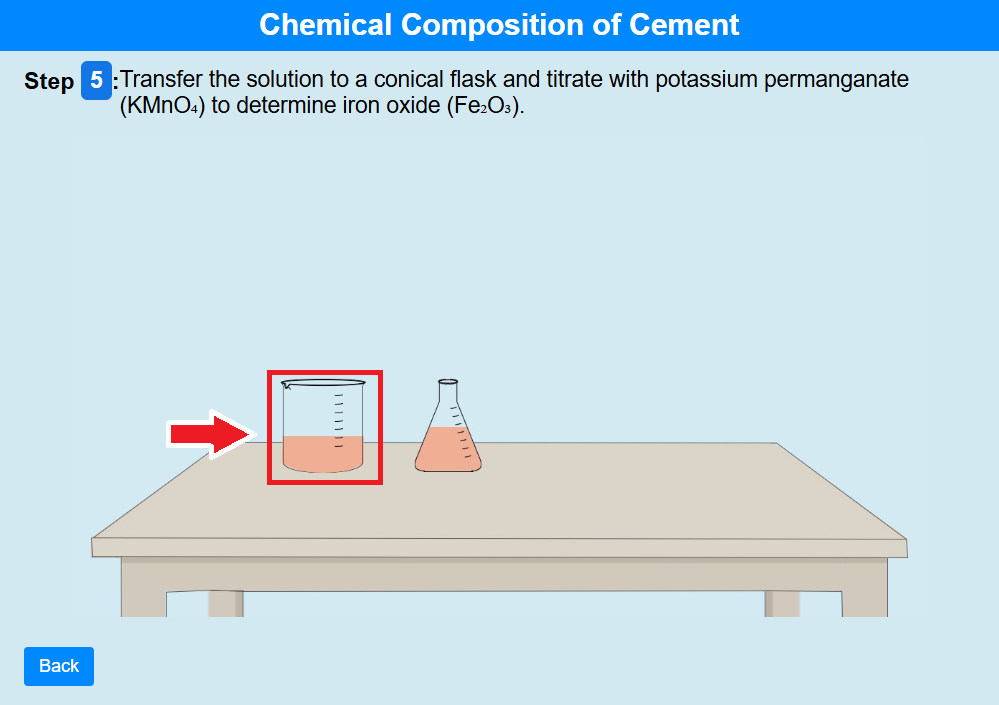
• Click on the knob to add The prepared solution in the conical flask is titrated with standard potassiumpermanganate (KMnO₄) solution.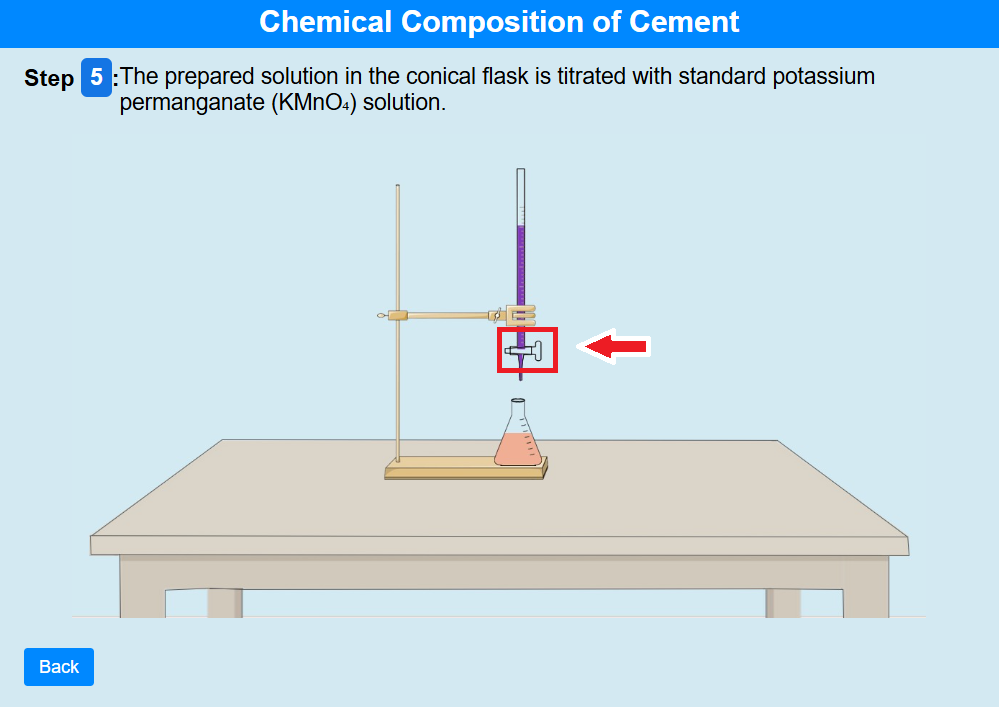
• Click on the knob to add. The titration is continued until a permanent light pink colour appears, indicating the end point. The KMnO₄ volume is noted to determine the Fe₂O₃% in the cement sample.
• Click on the calculate of Fe₂O₃% to calculate the percentage of Fe₂O₃%.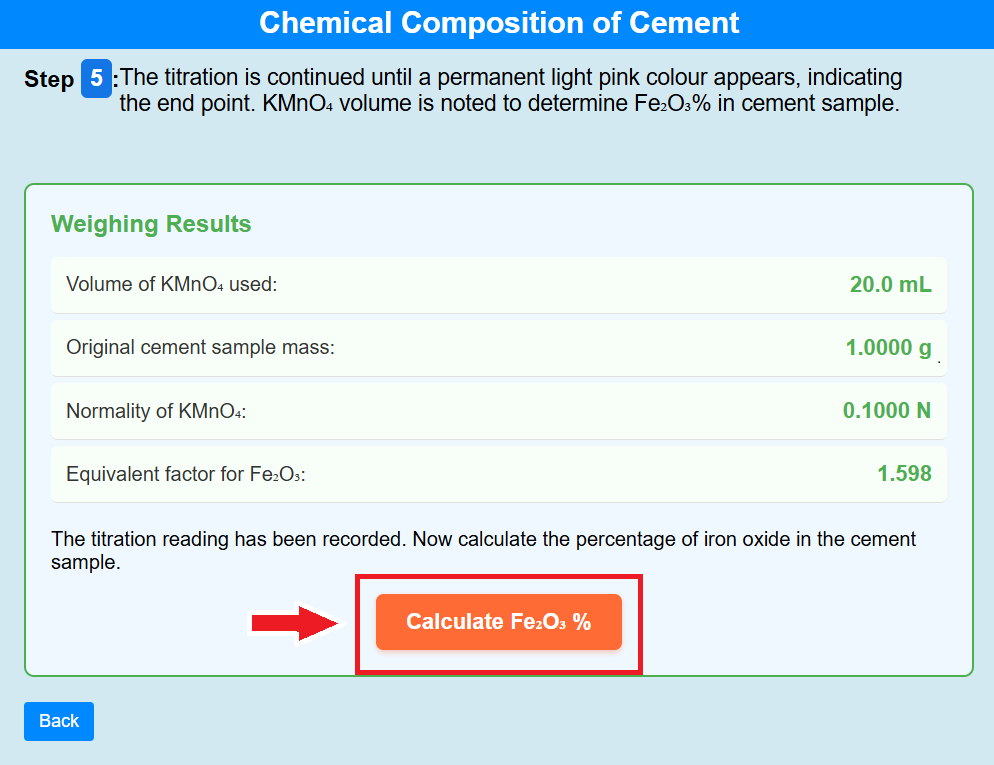
Step 6:
• Click on the burette to add the Sodium hydroxide (NaOH) solution, which is used to titrate the aluminium-containing solution.
• Click on the beaker to see aluminium react with sodium hydroxide to form aluminium hydroxide precipitate initially, which then dissolves in excess alkali, forming soluble aluminate.
• Click on the filter paper to filter the precipitate.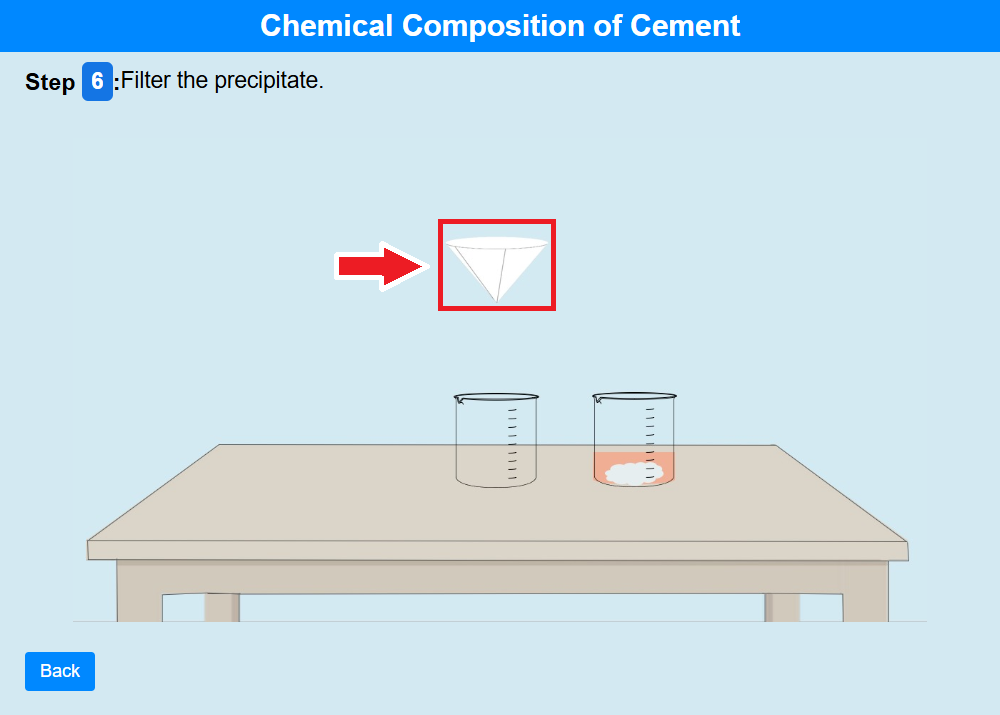
• Click on the filter paper to filter the precipitate.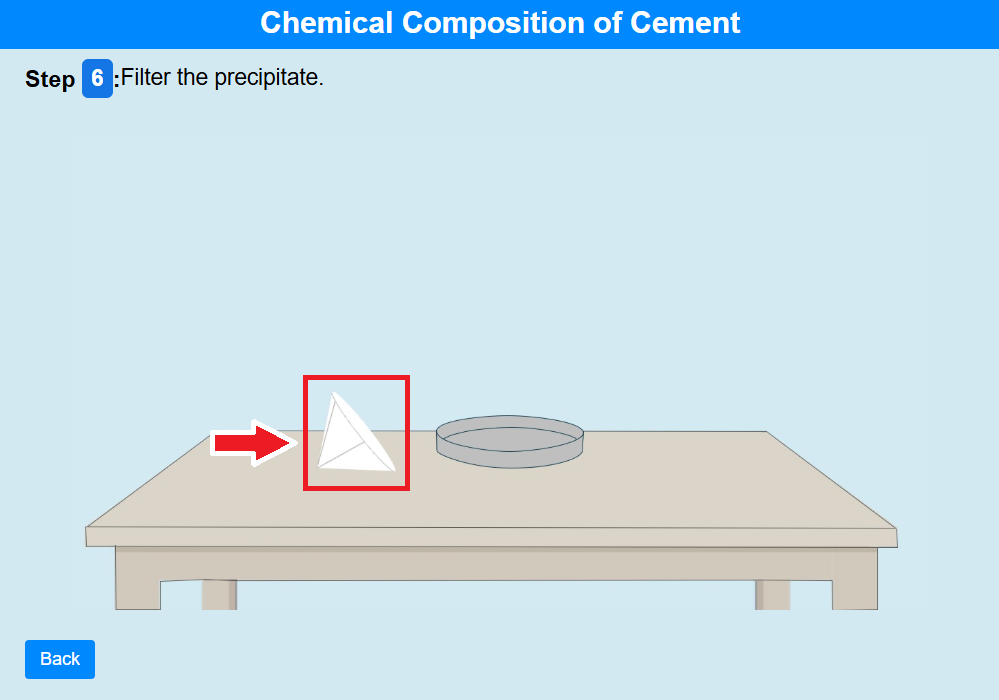
• Click on the knob of the oven to open it.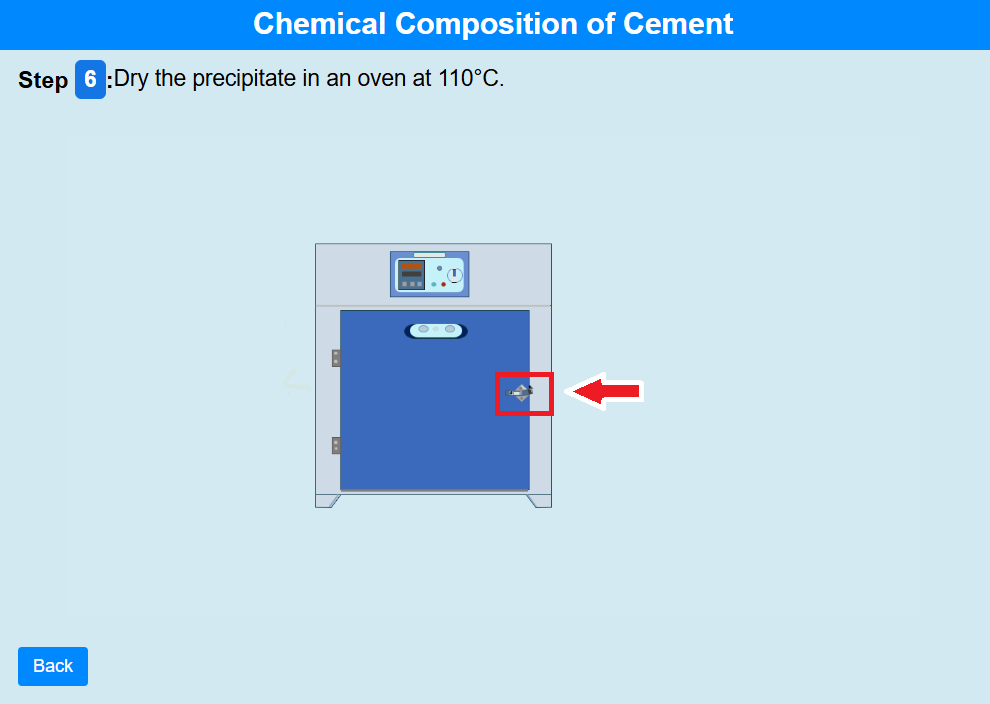
• Click on the crucible to dry it in an oven at 110°C.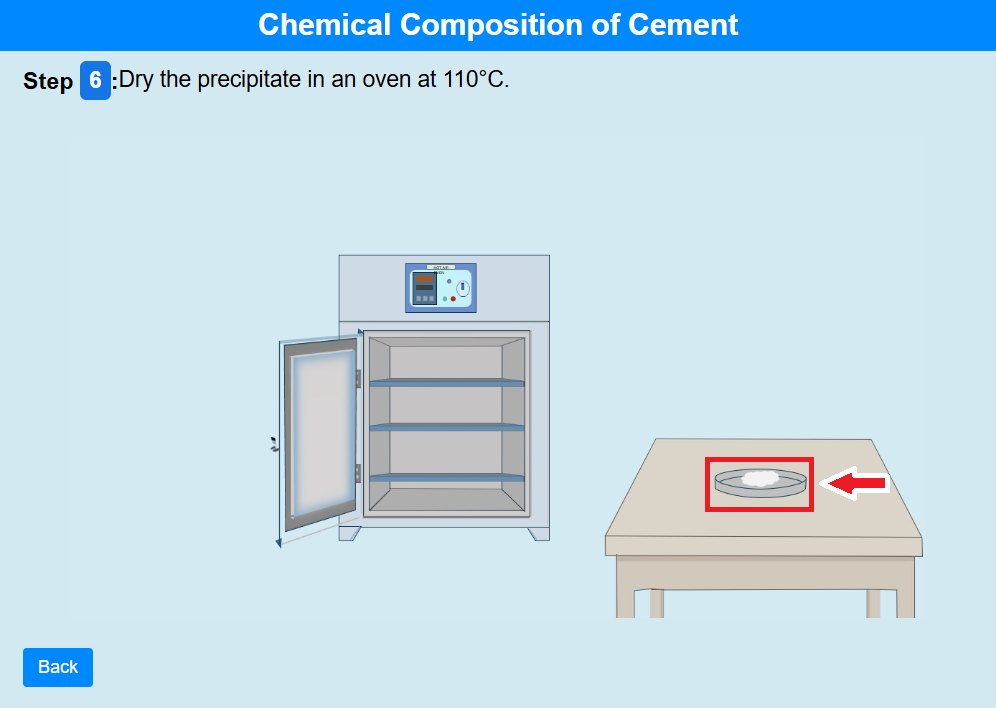
• Click on the oven screen and set the temperature to 110°C to dry.
• Click on the crucible to weigh the dried precipitate and use the titration data to calculate the percentage of aluminium oxide (Al₂O₃) present in the cement sample.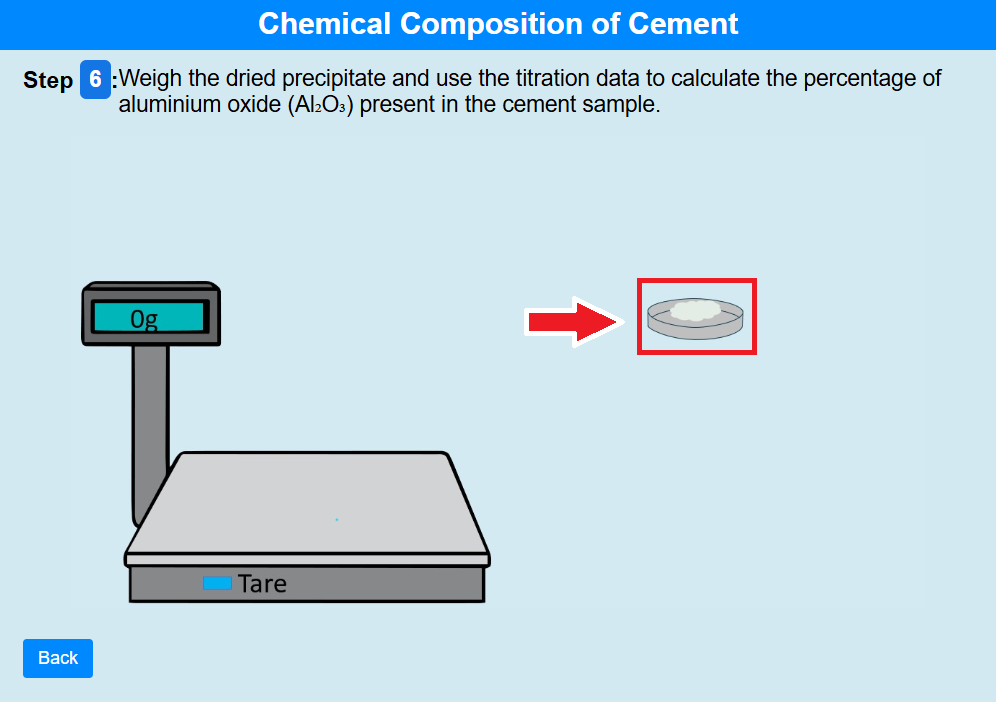
• Click on the calculate of Al₂O₃ % to calculate the percentage of Al₂O₃ %.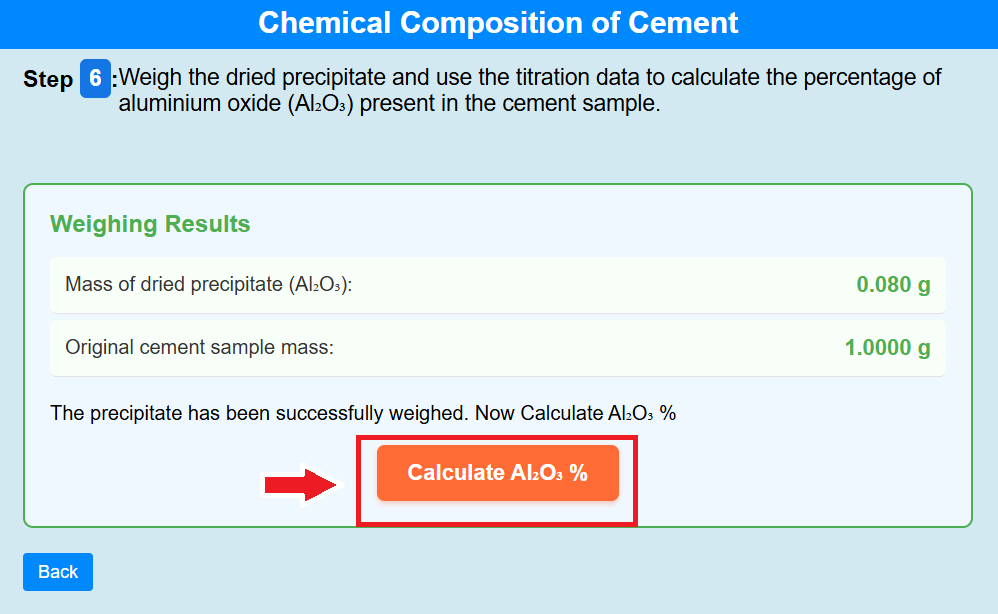
Determination of Magnesium Oxide
• Click on the link to perform the test of Determination of Calcium Oxide.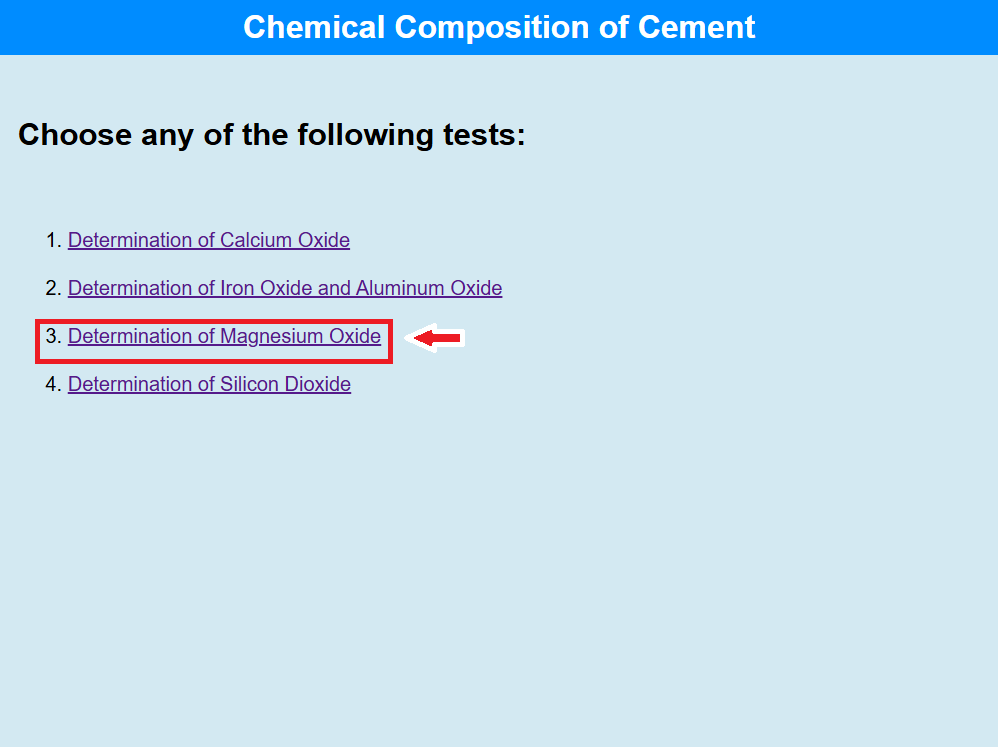
Step 1:
• Click on the pipette to use the remaining portion of the filtrate. Add diammonium phosphate
((NH₄)₂HPO₄) solution to the filtrate.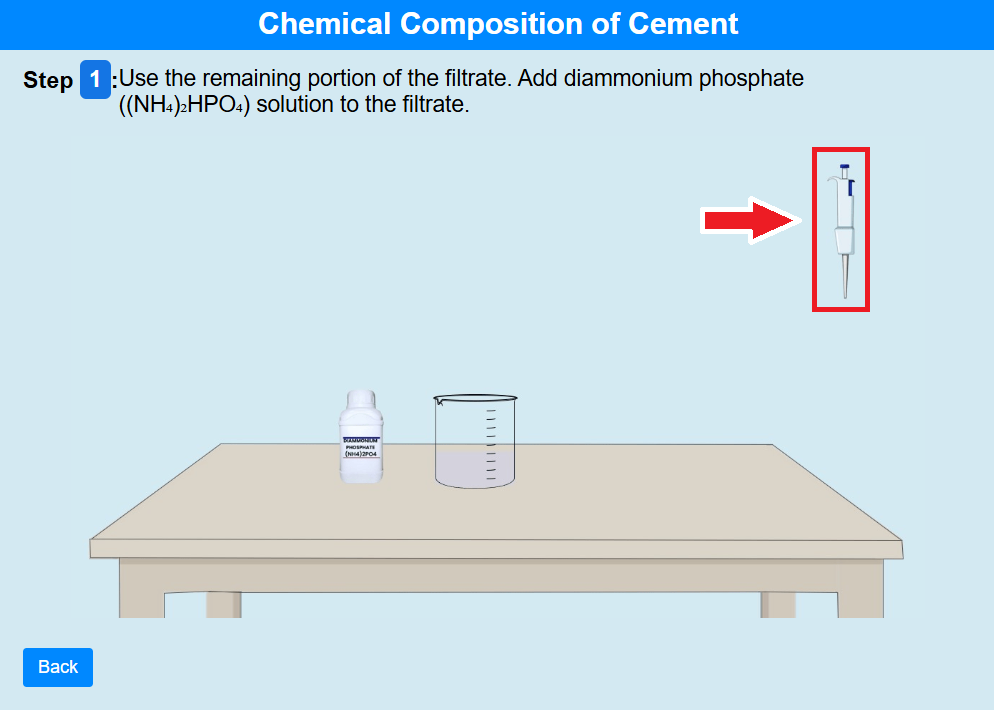
• Click on the beaker to observe the precipitate of magnesium ammonium phosphate (MgNH₄PO₄) is formed.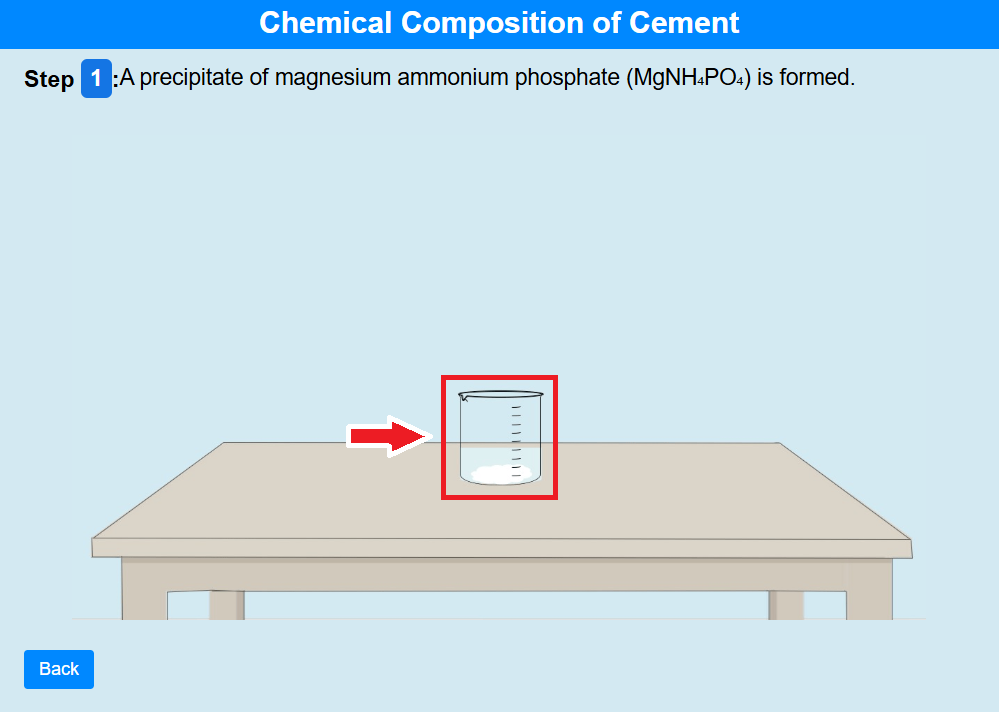
Step 2:
• Click on the filter paper to filter the precipitate.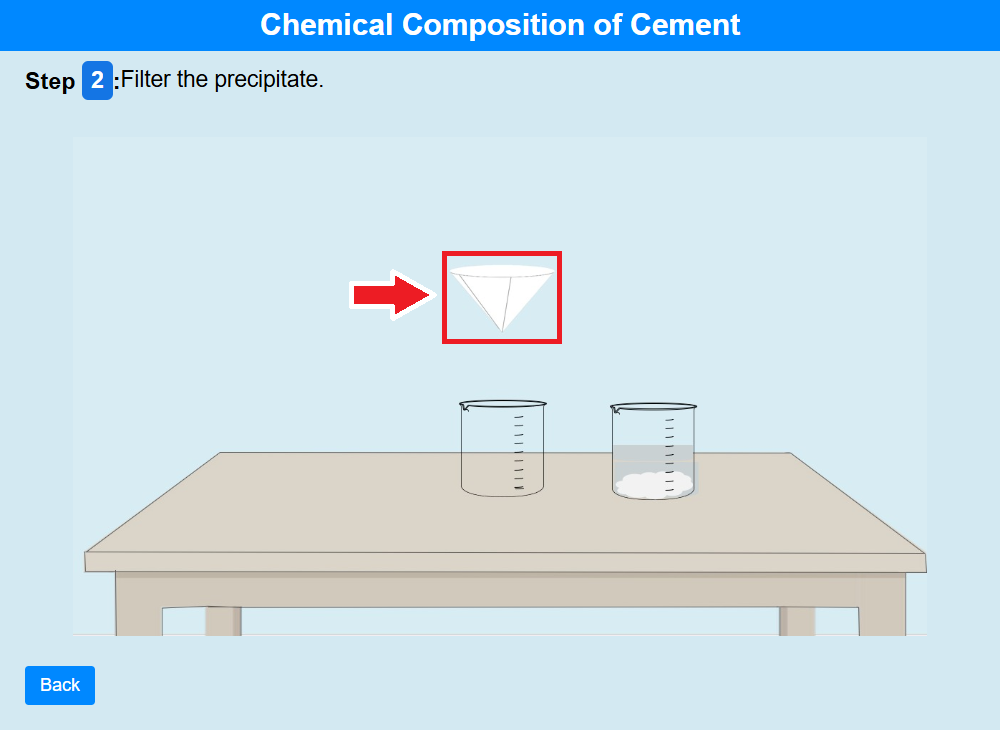
• Click on the filter paper. Transfer the filtrate to a clean crucible.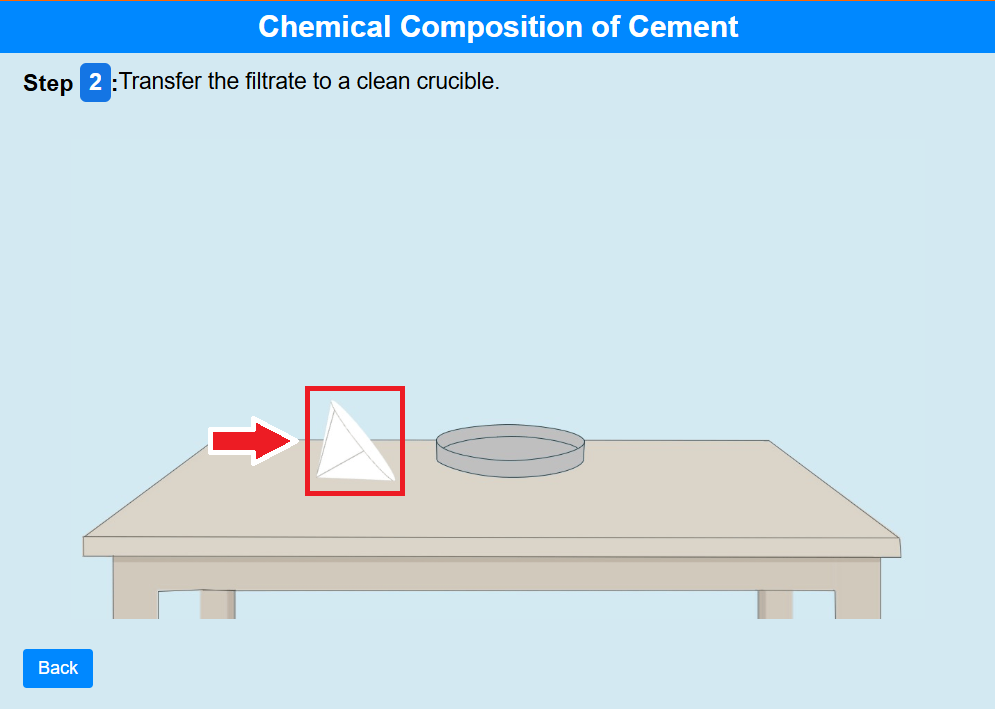
Step 3:
• Click on the crucible to dry the precipitate in an oven at 110°C.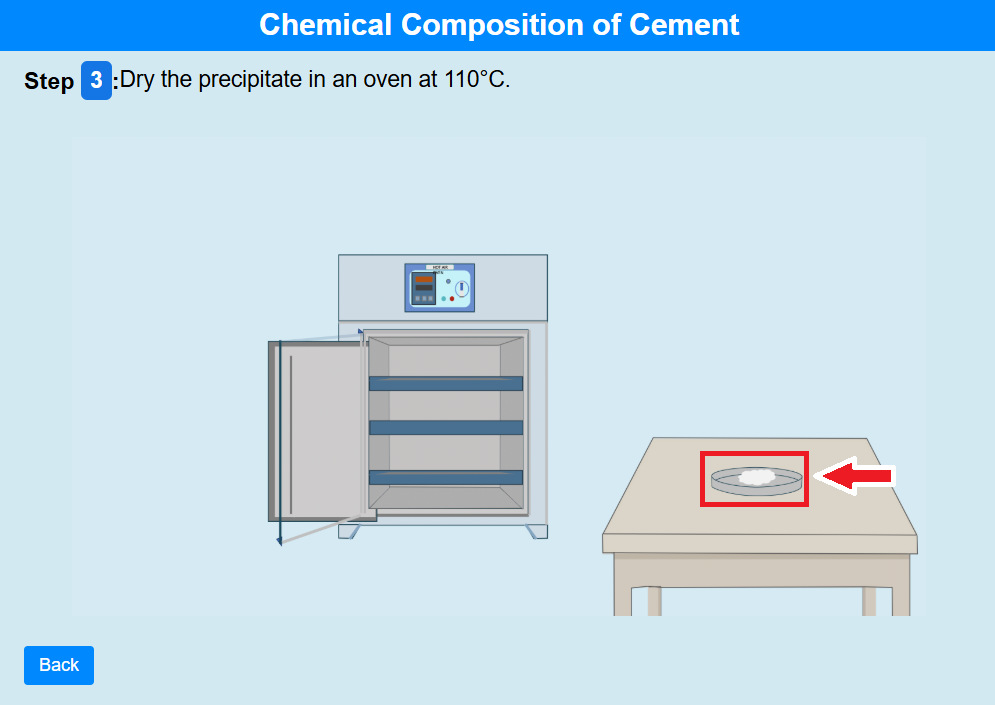
• Click on the screen to set the temperature.
Step 4:
• Click on the crucible to weigh the dried precipitate and use the weighing data to calculate the percentage of magnesium oxide (MgO) present in the cement sample.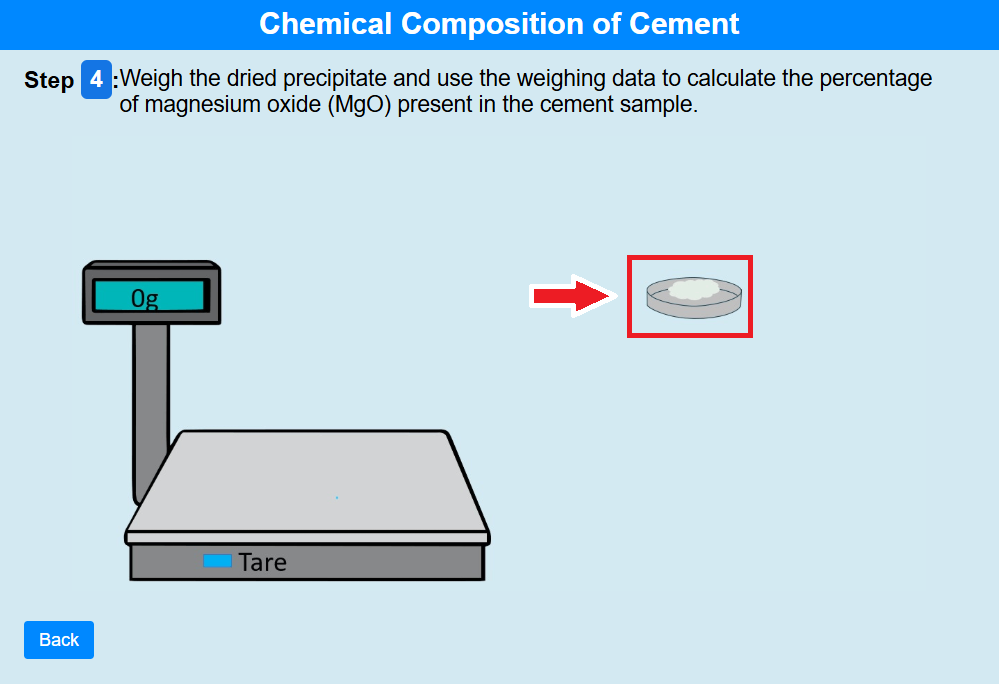
• Click on the calculate of MgO % to calculate the percentage of MgO%.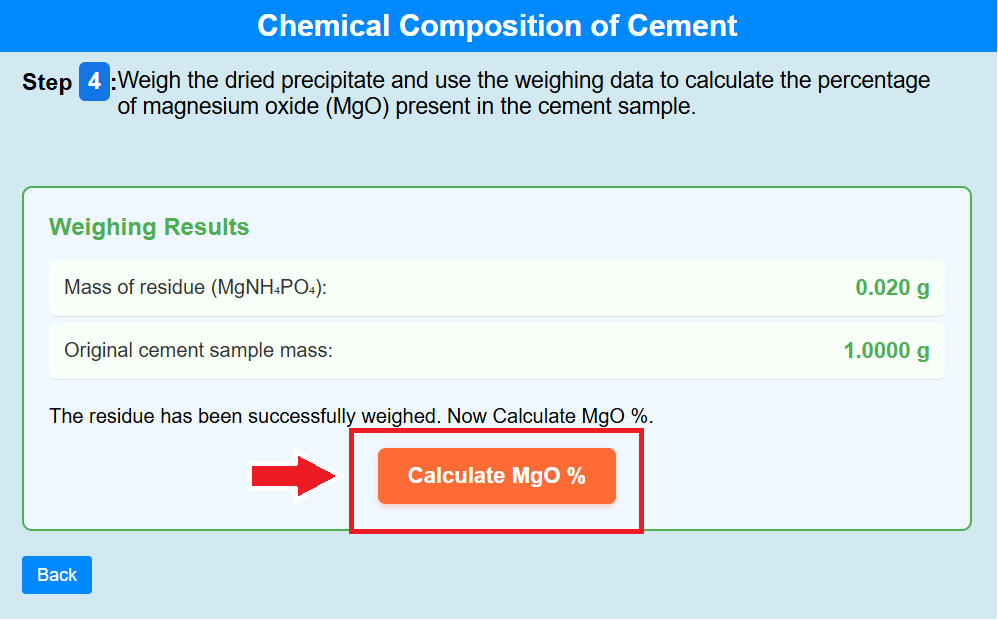
Determination of Silicon Dioxide
• Click on the link to perform the test of Determination of Calcium Oxide.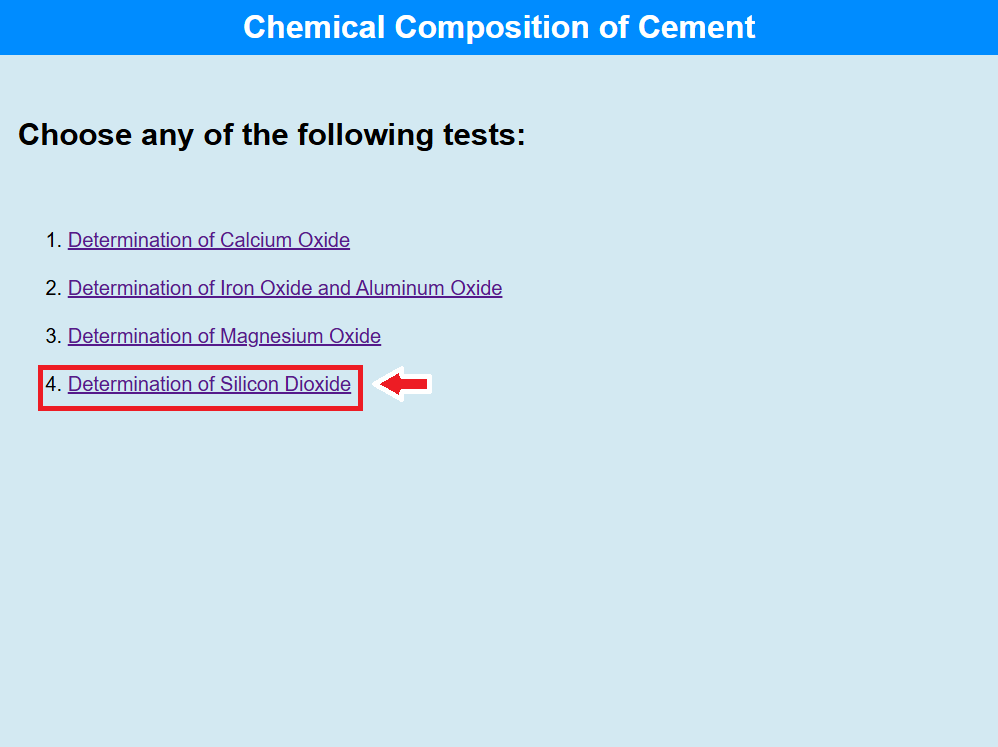
Step 1:
• Click on the pipette to take another portion of the filtrate into a beaker. Add dilute nitric acid (HNO₃) to it.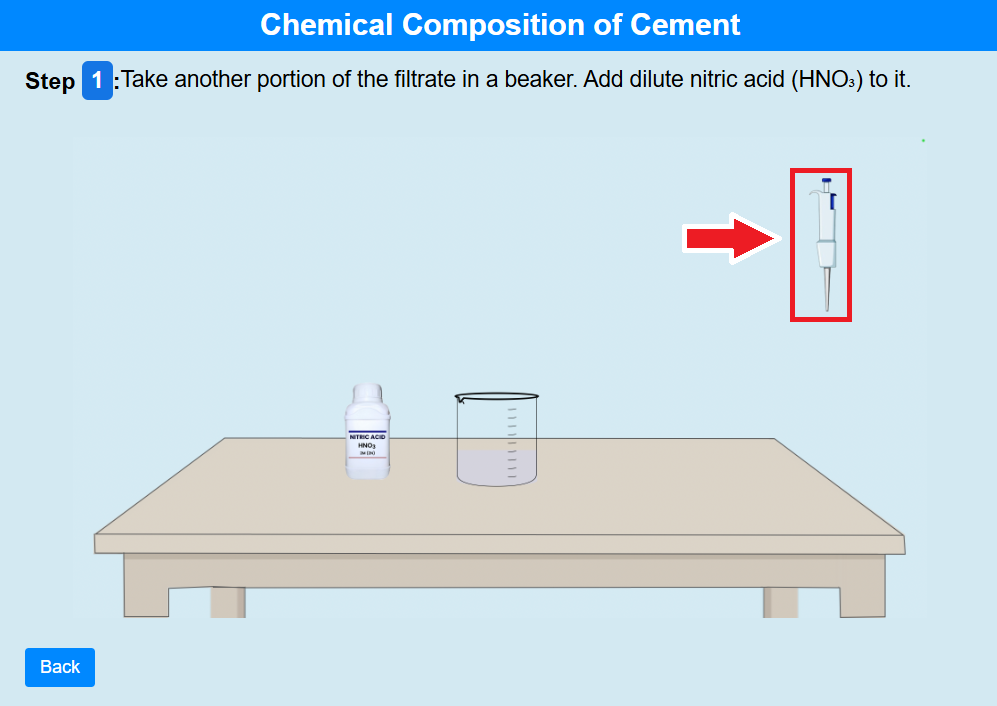
Step 2:
• Click on the beaker to heat the solution until it evaporates to dryness.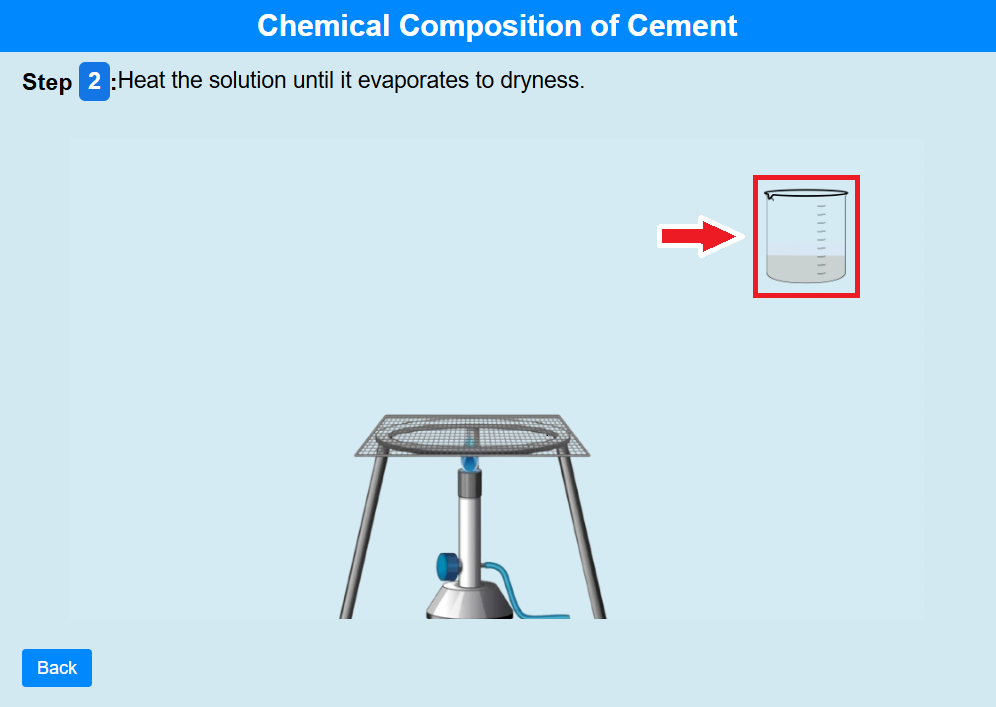
Step 3:
• Click on the filter paper to the residue obtained, represents silicon dioxide (SiO₂). Filter the precipitate and dry it in an oven at 110°C.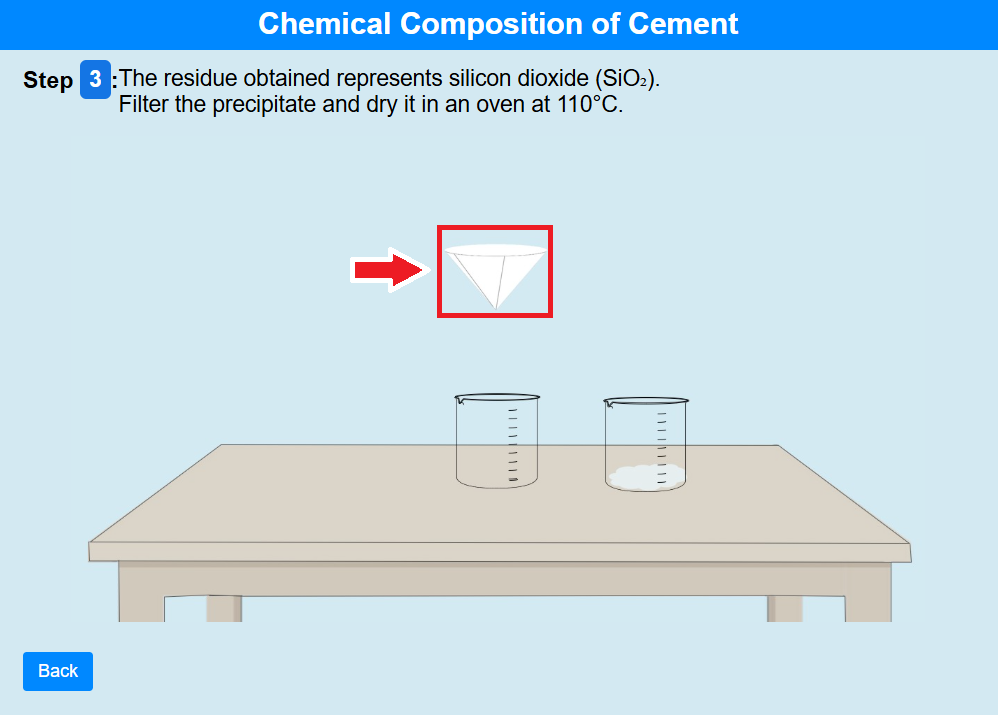
• Click on the filter paper to transfer the precipitate to a clean crucible.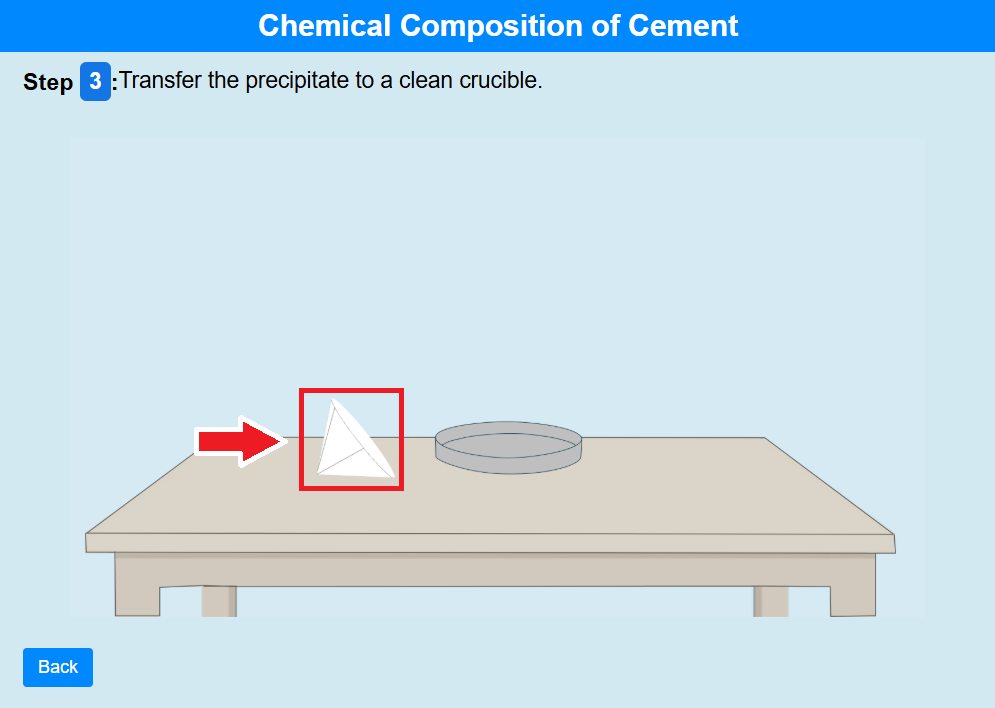
Step 4:
• Click on the oven knob to dry the precipitate in an oven at 110°C.
• Click on the crucible to heat the precipitate.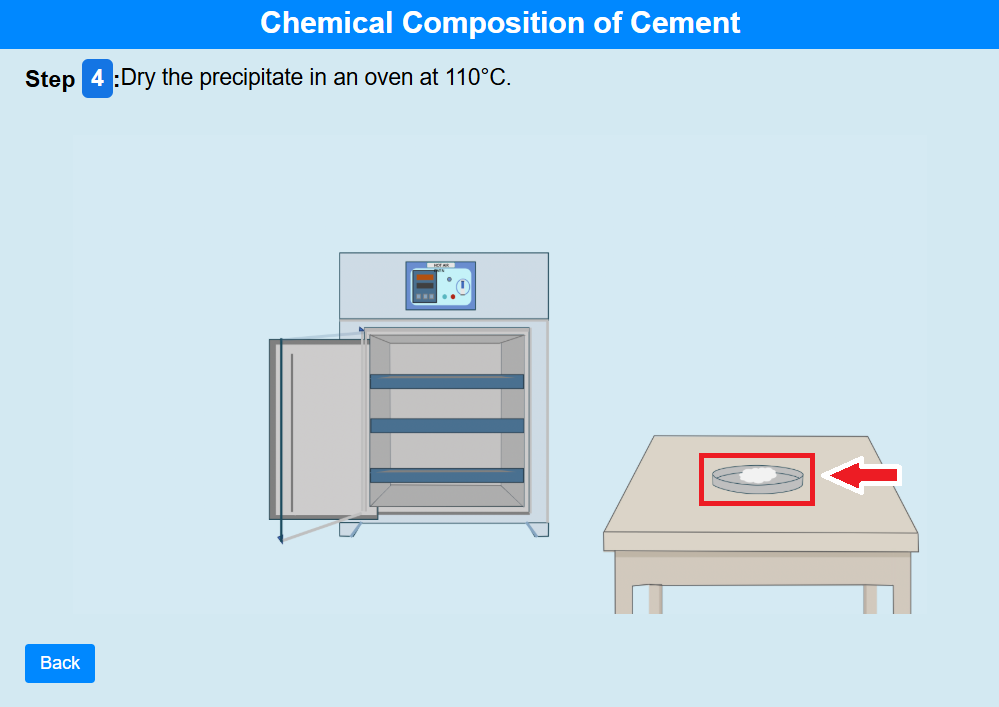
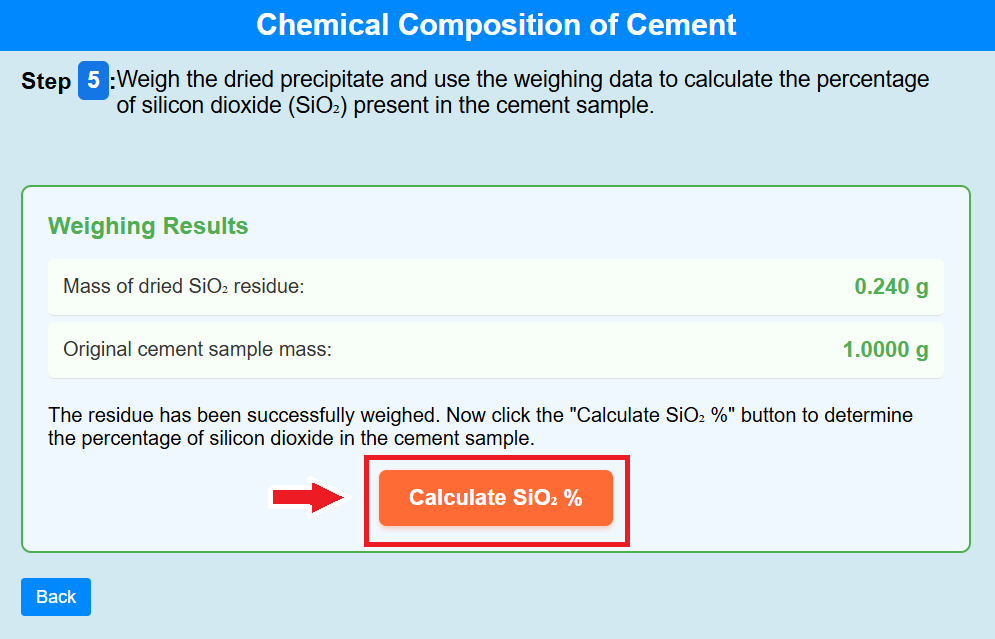
Step 5:
• Weigh the dried precipitate and use the weighing data to calculate the percentage
of silicon dioxide (SiO₂) present in the cement sample.
• Click on the calculate of SiO₂% to calculate the percentage of SiO₂%.