Rate constant and activation energy of potassium permanganate and oxalic acid reaction
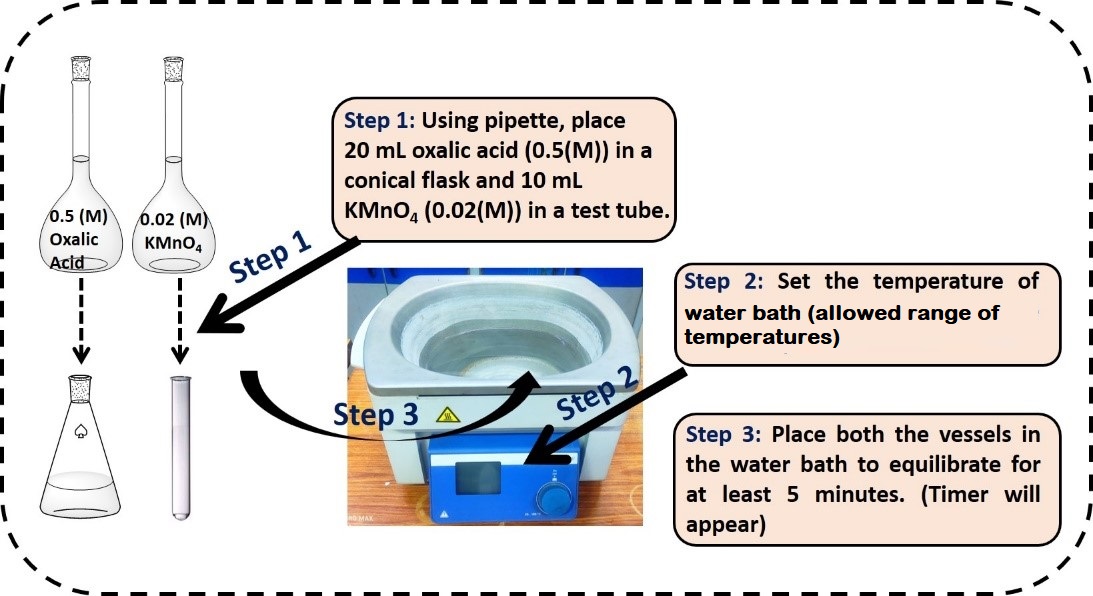
- Prepare 0.5 M oxalic acid and 0.02 M KMnO4 in a cleaned suitable volumetric flask. Pipette out 20 mL of oxalic acid and 10 ml of KMnO4 in a conical flask and a test tube respectively.
- Set the temperature of water bath up to the allowed range and place both the vessels into the water bath. Allow a 5 min equilibrium time for both of them.
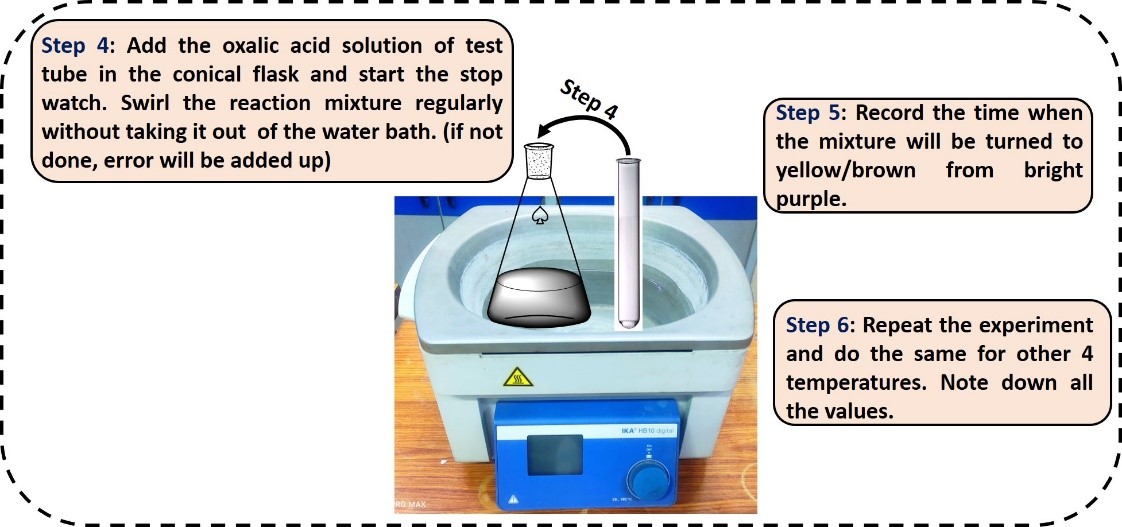
- Add the oxalic acid into the conical flask and start the stop watch to record time.
- Swirl the reaction mixture regularly keeping it to the water bath.
- Pause and record the time of the stop watch when the reaction mixture turned to purple from yellow/brown (completion of reaction).
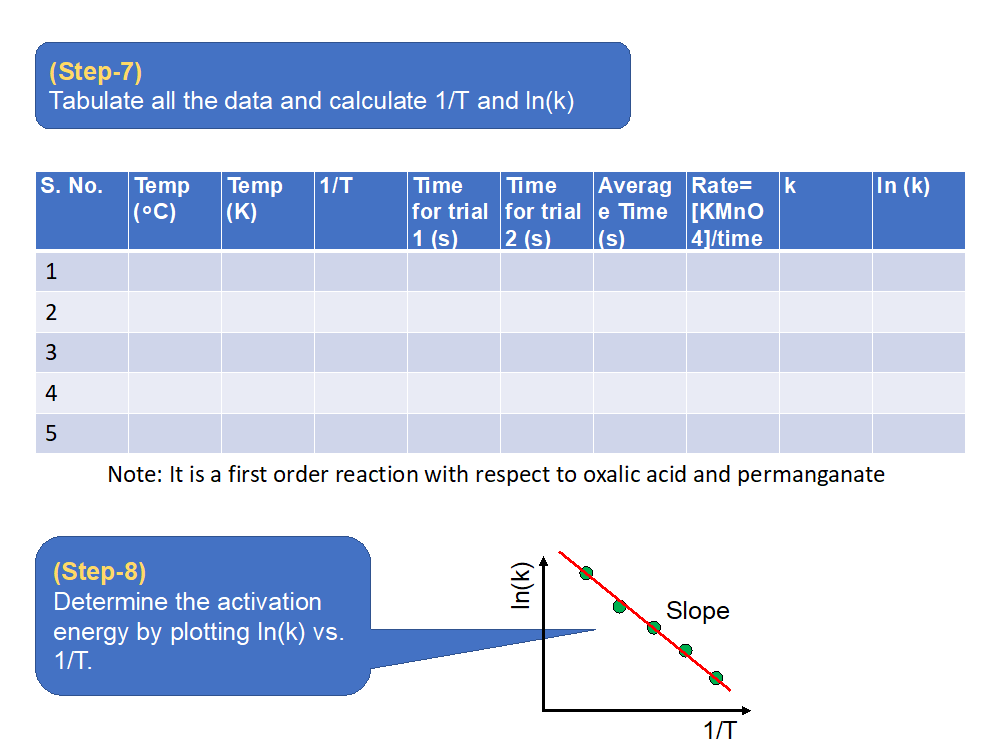
- Repeat the same procedure for 4 other temperatures and determine the activation energy of the reaction.
Materials & Reagents Required:
- Temperature controlled water bath.
- Volumetric flask (250 ml)
- Conical flask (250 ml)
- Test tubes
- Micropipette (5 ml)
Procedure in laboratory (diagram)
Procedure in laboratory

