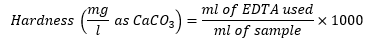Analysis of water for potable and food purposes
Water is an important ingredient used for drinking purpose and also in preparation of various food products. Quality of water plays important role in imparting desired properties to the processed products. Also, the drinking water is a good source of minerals but a limited amount is desired. The presence of different ions in water and their level affect the quality of water.
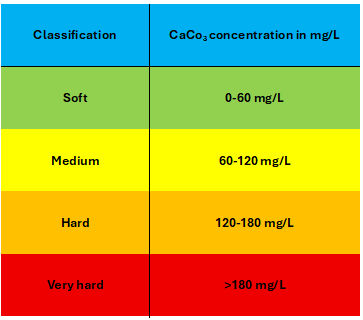
Water quality is analysed in different terms like hardness, chlorine content, chloride residue etc. Hardness of water is no specific constituent but is a variable and complex mixture of cations and anions. It is caused by dissolved polyvalent metallic ions. In fresh water, the principle hardness causing ions are calcium and magnesium. The other ions like Strontium, Iron, Barium and Manganese also contribute. Hardness is commonly expressed as CaCO3 in mg/L. The water can be classified into soft, medium hard and very hard water based on the concentration of CaCo3 as shown in the following diagram.
Ethylene diamine tetraacetic acid (EDTA) forms stable chelate complexes with calcium (Ca²⁺) and magnesium (Mg²⁺) ions in an alkaline medium (pH ≈ 10). In this titration, Eriochrome Black T (EBT) is used as a metal ion indicator. In the presence of free Ca²⁺ or Mg²⁺ ions, EBT forms a weak, wine-red (pink) colored complex. Since EDTA has a stronger affinity for these metal ions than the indicator, the metal ions preferentially bind to EDTA during titration. As EDTA is gradually added to the sample solution, it complexes with the free Ca²⁺ and Mg²⁺ ions, releasing the indicator into its unbound form. Once all the metal ions have been complexed by EDTA, the indicator remains free in solution and exhibits a blue color, indicating the endpoint of the titration. The amount of EDTA consumed, based on its known concentration and volume used, is then employed to calculate the concentration of calcium (or total hardness) in the sample, which is expressed as mg CaCO₃ per liter. It was calculated using the following formula.