Determination of Dissolved Oxygen in Water
Objective:
To estimate Dissolved Oxygen (DO) level in the given water sample by Winkler's Method.
Apparatus used:
BOD bottles, Pipette, Conical flask, Burette, Beaker, Dropper, Spatula etc.
Solutions used Manganese Sulphate solution, Alkali Iodide Azide solution, Concentrated Sulphuric Acid, Sodium Thiosulphate Solution, Potassium Iodide powder, Hydrochloric acid, Potassium Dichromate solution, Starch indicator DescriptionDissolved Oxygen is the amount of oxygen consumed by the organisms in breaking down the waste.
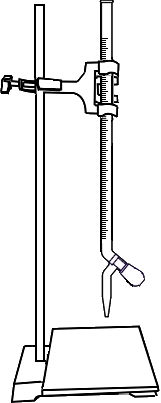





Fill a burette with Sodium Thiosulfate (Na2S2O3) solution.




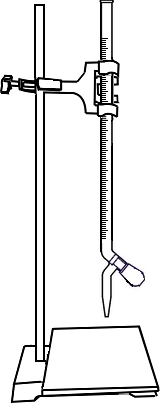



Take 10ml of 0.025N Potassium Dichromate (K2Cr2O7) in a conical flask.




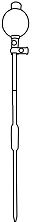












Add 2ml of Hydrochloric Acid (HCl) and 1 spatula of Potasium Iodide (KI) powder to the same conical flask.








Titrate the sample against Na2S2O3. Add 2ml Starch when the colour of the solution changes to pale yellow. Then continue the titration till blue colour becomes colourless.


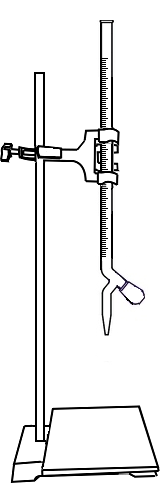




Open the knob to start the liquid running into the conical flask
Close the knob when the colour of solution in conical flask changes to pale yellow
Add 1ml Starch into conical flask
(The initial reading should be lower meniscus value) Initial burette reading = 0 ml
Observations:
| Normality of K2Cr2O7 solution (N) | Volume of K2Cr2O7 solution (ml) | Burette Reading (ml) | Volume of Na2S2O3 solution (ml) (Final Value - Initial Value) | |
| Initial Value | Final Value | |||
| 0.025 | 10 | |||
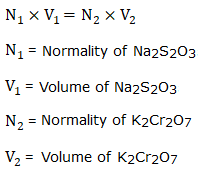
✔
✘
Normality of Na2S2O3 =
Determination of Dissolved Oxygen
Fill BOD bottle with sample water up to the bottle brim.












Immediately add 2ml of Manganese Sulfate (MnSO4) solution to the BOD bottle by inserting the automatic pipette just below the surface of the liquid.












Add 2ml of Alkali-Iodide-Azide reagent to the BOD bottle by inserting the automatic pipette just below the surface of the liquid.












Mix the sample by inverting BOD bottle several times and allow the precipitate to settle down at bottom.






Allow the precipitate to settle down for few minutes
Add 2ml of of concentrated Sulphuric Acid (H2SO4) via graduated pipette carefully without forming air bubbles.











Squeeze the pipette bulb and dip pipette into the H2SO4 solution
Press the up arrow on the bulb to take the liquid up into the pipette
Mix the sample by inverting the BOD bottle till all the precipitate dissolves.




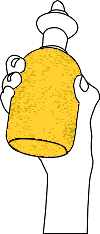
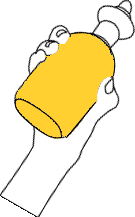

Take 200ml of sample in a conical flask using pipette.























For calculation purpose 200ml of sample is taken
Fill the burette with Sodium Thiosulfate (Na2S2O3) solution.









Titrate the sample against Na2S2O3. Add 2ml Starch when the colour of the solution changes to pale yellow. Then continue the titration till blue colour becomes colourless.











Open the knob to start the liquid running into the conical flask
Close the knob when the colour of solution in conical flask changes to pale yellow
Add 1ml Starch into conical flask
Initial burette reading = 0 ml
Observations:
| Volume of sample (ml) | Normality of Na2S2O3 solution (N) | Burette Reading (ml) | Volume of Na2S2O3 solution (ml) (Final Value - Initial Value) | |
| Initial Value | Final Value | |||
| 200 | ||||
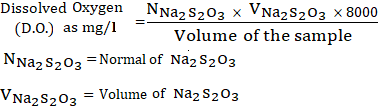
✔
✘
Dissolved Oxygen (mg/l) =
Inference:
What is the acceptable range of Dissolved Oxygen in drinking water?
0mg/l - 4mg/l 4mg/l - 7mg/l 7mg/l - 14mg/l >14mg/l